|
Getting your Trinity Audio player ready... |
Pharmacy Calculations: Understanding Density and Displacement Values
Introduction Density and Displacement Values
Density and Displacement Values Pharmacy calculations are an integral part of pharmaceutical practice, enabling professionals to ensure accuracy in formulations, drug dosages, and the preparation of medications. Among the fundamental concepts that pharmacy students must master are density and displacement values. These principles have significant implications in pharmaceutical calculations, affecting how we measure and mix substances.
Density, a term commonly used in chemistry and pharmacology, describes the relationship between the mass of a substance and the volume it occupies. This relationship is crucial when dealing with different types of materials, including solids, liquids, and semi-solids, in the pharmaceutical field. The principle of displacement is closely related, particularly when dealing with powders, solutes, or compounds that can alter the volume of a solution when dissolved.
This section will explore both density and displacement values in depth, providing several key examples of how these concepts are applied in pharmaceutical calculations. These examples will help pharmacy students and professionals grasp how to navigate situations that involve calculating mass, volume, and concentrations based on density and displacement principles.
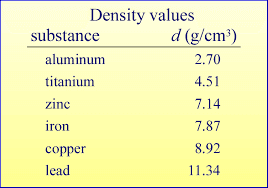
What is Density?
At its core, density is a measure of how much mass a substance has in a given volume. It is commonly expressed as the ratio of mass to volume, typically in units of grams per milliliter (g/mL) for liquids or grams per cubic centimeter (g/cm³) for solids. This value is essential for the accurate formulation and preparation of pharmaceutical products, particularly when the ingredients or substances used have varying densities.
To put it into perspective, let’s consider an example using water. At a temperature of 20°C, 100 grams of water will occupy 100 milliliters of space. This creates a 1:1 ratio of mass to volume. However, different substances do not exhibit this same proportional relationship. For instance, a substance like glycerin is much denser than water, meaning the same volume of glycerin will weigh more than the equivalent volume of water.
When working with liquids, solids, or semi-solids, understanding the density is essential because it allows formulators to convert between mass and volume. This is particularly important when dealing with ingredients that are easier to measure by volume rather than mass, or vice versa.
Example 1: Conversion Between Volume and Mass
Consider the following scenario:
- You are given 37.5 mL of Liquid X and 18.2g of Liquid Y.
- The density of Liquid X is 0.65 g/mL, and the density of Liquid Y is 1.1 g/mL.
To find the mass of Liquid X, multiply the volume by its density:
Mass of Liquid X=37.5 mL×0.65 g/mL=24.4 g\text{Mass of Liquid X} = 37.5 \, \text{mL} \times 0.65 \, \text{g/mL} = 24.4 \, \text{g}
For Liquid Y, to find the volume it occupies, divide its mass by its density:
Volume of Liquid Y=18.2 g1.1 g/mL=16.5 mL\text{Volume of Liquid Y} = \frac{18.2 \, \text{g}}{1.1 \, \text{g/mL}} = 16.5 \, \text{mL}
This shows that 37.5 mL of Liquid X has a mass of 24.4 grams, and 18.2 grams of Liquid Y occupies 16.5 mL.
These conversions are fundamental for pharmacists and formulators because they enable them to measure liquids and solids accurately and adjust formulations accordingly, ensuring the correct dosage and consistency in pharmaceutical products.
Example 2: Ointment Preparation
Consider the excipients for preparing an ointment:
- Liquid paraffin: 20 mL
- Zinc oxide: 15 mg
- Yellow soft paraffin to: 100g
The goal is to calculate the final formula for 25g of ointment.
Since liquid paraffin has a density of 0.88 g/mL, we multiply the volume by the density to find the mass:
Mass of Liquid Paraffin=20 mL×0.88 g/mL=17.6 g\text{Mass of Liquid Paraffin} = 20 \, \text{mL} \times 0.88 \, \text{g/mL} = 17.6 \, \text{g}
Now, let’s break down the quantities for the final formula. If the total ointment mass must be 25g, the remaining mass after accounting for liquid paraffin and zinc oxide is:
25 g−(17.6 g+0.015 g)=7.4 g25 \, \text{g} – (17.6 \, \text{g} + 0.015 \, \text{g}) = 7.4 \, \text{g}
This remaining 7.4 grams will be composed of yellow soft paraffin, which is calculated by adjusting proportions based on the weights and the density of the ingredients.
Displacement Values
In pharmaceutical formulations, displacement refers to the volume occupied by a substance when it is dissolved in a liquid or mixed with other substances. The concept of displacement volume becomes especially important when dealing with powders or solutes that affect the overall volume of the solution or mixture. Understanding displacement values allows formulators to calculate the correct volumes of solvents and diluents needed to maintain the correct concentration and potency of a medication.
Example 3: Calculating the Volume of Water for Injection (WFI)
Let’s consider a drug with a displacement volume of 0.07 mL/5mg. If we need to prepare 1 mL of injection containing 5mg of drug X, the volume of Water for Injection (WFI) required can be calculated as follows:
First, we know that 5 mg of drug X displaces 0.07 mL. To achieve a total volume of 1 mL, we subtract the displacement volume from the final volume:
Volume of WFI=1.0 mL−0.07 mL=0.93 mL\text{Volume of WFI} = 1.0 \, \text{mL} – 0.07 \, \text{mL} = 0.93 \, \text{mL}
Thus, 0.93 mL of WFI is needed to make 1 mL of injection containing 5 mg of drug X.
Example 4: Diluent Calculation
Now, let’s calculate the required volume of a diluent to prepare an injection with a concentration of 4 mg/mL. If the displacement volume of the drug is 0.5 mL/40 mg, we can follow a similar process:
- Determine the final volume: For 80 mg of drug X, the required volume is:
Required volume=80 mg4 mg/mL=20 mL\text{Required volume} = \frac{80 \, \text{mg}}{4 \, \text{mg/mL}} = 20 \, \text{mL}
- Determine displacement volume: For 80 mg, the displacement volume is:
Displacement volume=80 mg40 mg×0.5 mL=1 mL\text{Displacement volume} = \frac{80 \, \text{mg}}{40 \, \text{mg}} \times 0.5 \, \text{mL} = 1 \, \text{mL}
- Subtract displacement from final volume:
Volume of diluent=20 mL−1 mL=19 mL\text{Volume of diluent} = 20 \, \text{mL} – 1 \, \text{mL} = 19 \, \text{mL}
Thus, 19 mL of diluent is needed to prepare a concentration of 4 mg/mL.
Conclusion
Both density and displacement values are crucial concepts in pharmacy calculations, particularly when preparing formulations, calculating drug doses, and ensuring the accuracy of pharmaceutical products. By mastering these principles, pharmacy students and professionals can navigate a wide range of calculation challenges effectively. These concepts are essential not only for drug formulation but also for understanding how substances behave when combined in solution.
As demonstrated in the examples, understanding the relationship between mass, volume, and displacement enables accurate measurements and proper formulation adjustments. With practice, pharmacy students can gain confidence in applying these calculations in real-world scenarios, ensuring that medications are prepared safely and effectively for patients. Ultimately, precision in pharmacy calculations translates to better patient outcomes and the success of pharmaceutical therapies.
Summary of Key Concepts
| Concept | Explanation |
|---|---|
| Density | A measure of the mass of a substance per unit volume. Critical for converting between mass and volume. |
| Displacement Volume | The volume occupied by a substance when dissolved or mixed with others. Important for accurate formulations. |
| Formula for Density | Density=MassVolume\text{Density} = \frac{\text{Mass}}{\text{Volume}} |
| Practical Application | Used to adjust volumes or masses of ingredients in pharmaceutical products, such as injections, ointments, and suspensions. |