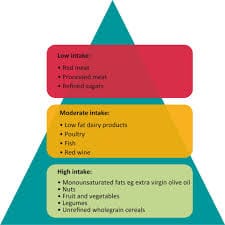
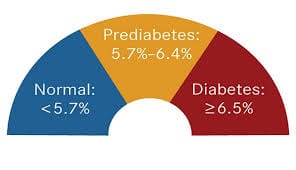
CMC regulatory affairs: An introduction to CMC and compliance V5.0
CMC regulatory affairs: An introduction to CMC and compliance CMC regulatory affairs CMC Regulatory affairs is an important component of…
eCTD Regulatory affair & ICH-GCP-V4.0
eCTD Regulatory affair & ICH-GCP eCTD Regulatory affair & ICH-GCP Regulatory affairs is a critical component of clinical research and…
Basaglar and Toujeo-Basaglar is approved for use in adults and children (ages 6 years and older)
Basaglar and Toujeo are both long-acting insulin formulations used to treat diabetes. Basaglar is a long-acting insulin-Basaglar and Toujeo Basaglar…
AstraZeneca has acknowledged that its vaccine may lead to clotting, but there\’s no need for panic among Covishield vaccine recipients in India.
AstraZeneca has acknowledged that its vaccine may lead to clotting, but there\’s no need for panic among Covishield vaccine recipients…
Module 5 clinical summary report regulatory submission
Module 5 clinical summary report regulatory submission module 5 on clinical summary reports. Module 5 clinical summary report regulatory submission…
Non-clinical Regulatory affair module 4
Non-clinical Regulatory affair module 4 Non-clinical Regulatory affair module 4 Regulatory non-clinical Regulatory Affairs Non-Clinical Module 4 is an essential…
eCTD-module-3-drug-products in eCTD/CTD/ACTD Format-Part 2
eCTD-module-3-drug-products Understanding the 5 Modules of Regulatory Affairs in eCTD/CTD/ACTD Format-Part 2 eCTD-module-3-drug-products Module 3 drug products eCTD-module-3-drug-products Module 3…
Understanding the 5 Modules of Regulatory Affairs in eCTD/CTD/ACTD Format-Part 1
Understanding the 5 Modules of Regulatory Affairs in eCTD/CTD/ACTD Format-Part 1 Regulatory Affairs Module 1-Understanding the 5 Modules Understanding the…
Regulatory Affairs Requirement-(V24) for submission dossier
Regulatory Affairs Requirement for submission dossier Regulatory Affairs Requirement: USFDA Regulatory Affairs Requirement The United States Food and Drug Administration…
Clinical Study Report-E3 structure
Clinical Study Report-E3 structure Clinical Study Report First, is important to understand the definition, requirements, and potential uses of a…