|
Getting your Trinity Audio player ready... |
Process Requirements for Film Coating Systems
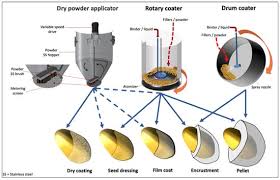
Process Requirements for Film Coating Systems Film coating is a critical process in the pharmaceutical industry, especially when it comes to producing tablets with a uniform and reliable coating. The success of the film coating process is determined by several factors, and understanding these requirements ensures that the coating is applied evenly, maintains its integrity, and meets the quality standards required for pharmaceutical products.
An effective coating should be thick, smooth, and free from defects like cracks or mechanical damages. To achieve this, there are four main process requirements for film coating systems that must be meticulously controlled and optimized. These include the adequacy of spray atomization, proper mixing and agitation of tablets, the correct input of heated air, and the presence of a good exhaust facility.
1. Adequate Spray Atomizing Means: Process Requirements for Film Coating Systems
One of the most important steps in film coating is the atomization of the coating liquid. The spray should be fine enough to evenly distribute the liquid over the tablet core. This atomization is critical because it determines how the coating solution interacts with the tablet surface, affecting the uniformity and quality of the film. The efficiency of this step hinges on the precise control of several factors, including the rate of spray and the air pressure used during the spraying process.
The spraying rate plays a significant role in determining the coating’s quality. If the rate is too low, the coating liquid may not wet the tablet surface adequately, leading to incomplete coverage. On the other hand, spraying at a high rate can result in excessive wetting, which can cause defects like picking or stacking, where tablets stick to each other or to the equipment. The goal is to maintain an optimal spray rate, one that ensures proper wetting without over-wetting the tablets.
Air pressure is another critical parameter. When the spray air pressure is too high, it can create very fine droplets, which may not adequately cover the tablet surface and can lead to coalescence or the pooling of the coating solution in certain areas. In contrast, insufficient air pressure may cause the formation of larger droplets, which could lead to uneven coating, sticking, and tablet clumping.
Therefore, controlling spray atomization—by adjusting both the air pressure and the spray rate—is essential for achieving a smooth and uniform coating. This not only helps in applying the coating evenly but also minimizes defects and ensures the coating solution reaches the tablets as required.
2. Proper Mixing and Agitation of the Tablets
The mixing and agitation of the tablets during the coating process are critical for ensuring uniformity. The speed of the rotating tablet pan plays a key role in how well the tablets are mixed with the coating solution. A higher rotational speed leads to more even distribution of the coating across all tablets by reducing any variations in the coating thickness.
The rotation speed determines the amount of time each tablet spends in the spraying zone, and this, in turn, influences the consistency of the coating on every tablet in the batch. While increasing the speed can help improve uniformity, excessive rotation can cause mechanical damage to the tablets, such as breakage or chipping, especially for tablets that are more fragile. Therefore, an optimal balance must be struck to promote efficient mixing while minimizing the risk of tablet damage.
The agitated mixing process also ensures that the coating solution is distributed evenly, helping the tablets maintain consistent coating thickness. Additionally, if the tablet pan is equipped with a mechanism to ensure consistent mixing, it will prevent tablet clustering and facilitate even drying after the application of the coating solution.
3. Sufficient Energy Input for Heated Air
In the film coating process, air is used not only to assist in the atomization of the coating solution but also to facilitate the drying of the coating once it has been applied. This drying step is crucial for ensuring that the film sets properly and that excess moisture is removed from the tablets. The temperature of the heated air plays a significant role in this process, and its adjustment can have a profound effect on both the efficiency of the drying process and the final quality of the coated tablets.
Increasing the temperature of the inlet air can speed up the drying process, as the warm air will help evaporate the solvent or water in the coating solution. This leads to faster film formation around the tablet. However, too much heat can cause premature drying, where the coating starts to dry before it fully covers the tablet, resulting in an uneven or incomplete coating. Excessive temperatures can also weaken the film’s integrity, making it prone to cracking.
On the other hand, insufficient air temperature will slow down the drying process, potentially causing the tablets to remain overly wet and susceptible to issues like smudging or the formation of a poor-quality coating. Maintaining the right balance of temperature and humidity is essential to ensure the uniformity of the coating and the overall quality of the tablets.
A well-calibrated air temperature system ensures that the drying process progresses at the right pace, preventing problems like over-wetting and under-drying, which can negatively impact the appearance and efficacy of the final product.
4. Effective Exhaust System
An efficient exhaust system is another key component of a successful film coating process. The exhaust facility serves multiple purposes, including removing dust particles and solvent-laden air from the coating chamber. The removal of these airborne particles is crucial not only for maintaining a clean environment but also for preventing contamination that could affect the quality of the tablets. Furthermore, proper ventilation helps prevent the buildup of volatile solvents, which could lead to the formation of potentially dangerous fumes.
By ensuring that the coating system is well-ventilated, you can maintain the cleanliness of the equipment, reduce the chances of contamination, and avoid any safety issues related to the release of harmful gases. A good exhaust system also ensures that the working environment remains safe for operators and that the process continues smoothly without any interruptions.
Types of Drugs Suitable for Film Coating
Film coating is widely used for many types of pharmaceutical drugs, especially tablets, to improve their appearance, ease of swallowing, and stability. Examples of drugs that are commonly film-coated include Valsartan 320 mg, Ranitidine 300 mg, Ondansetron 8 mg, Zinc 30 mg, Ibuprofen 200 mg, Atenolol 100 mg, and Accord Clarithromycin 500 mg. These drugs benefit from the protective and functional properties provided by the coating, which can also control the release of the drug or mask unpleasant tastes and smells.
Aqueous vs. Solvent-Based Film Coating: A Comparison
Film coating processes can be broadly divided into two categories: aqueous film coating and solvent-based film coating. Each approach has its unique benefits and challenges.
- Aqueous Film Coating: This method uses water as the primary solvent for the coating solution. Aqueous coatings are non-toxic and pose fewer health risks to operators, making them safer to work with. However, because water-based coatings take longer to dry, they require more extensive drying processes and increased ventilation to ensure the evaporation of the solvent. Aqueous coatings also tend to be slower to set compared to solvent-based coatings, which may result in slower production rates.
- Solvent-Based Film Coating: Solvent-based coatings use organic solvents such as methanol to dissolve the coating material. These coatings generally dry much faster, as the solvent evaporates quickly. This makes the process more efficient and allows for faster production. However, solvent-based coatings can pose health risks due to the toxicity of the solvents used, and the production area must be well-ventilated to minimize exposure. Additionally, the solvents can sometimes affect the tablet’s smell or taste, which could be a concern for some drug formulations.
Both methods have their place in pharmaceutical manufacturing, but the choice between aqueous and solvent-based coating depends on factors such as the type of drug being coated, production speed requirements, and safety considerations.
Quality Standards for Film Coating Systems
For any film coating system to function efficiently and produce high-quality results, it must meet certain global standards. These standards ensure that the equipment is built to operate safely, reliably, and in compliance with industry regulations. Recommended quality standards for film coating systems include:
- FDA Licensing: The system must be FDA-approved to ensure that it meets all necessary regulatory requirements for pharmaceutical production.
- GMP Certification: The system should be GMP (Good Manufacturing Practice) certified, guaranteeing that it adheres to the required practices for producing safe and effective pharmaceutical products.
- ISO Certification: The system should also have ISO 9001:2000 certification, ensuring that the manufacturer follows established international standards for quality management.
- Halal and Kosher Certification: These certifications may be required for certain products to meet dietary or religious needs.
By adhering to these certifications and standards, pharmaceutical manufacturers can ensure that their film coating systems are capable of producing high-quality, safe, and effective products.